Which Products Will Not Have a Digital Product Passport (DPP) – and Why It Matters for Business


The Digital Product Passport (Digital Product Passport – DPP) will be a key instrument of the new European framework for sustainable products (ESPR).
But not all goods will be required to have a DPP.
Food, animal feed, medicines, veterinary products, live plants and animals, as well as products of human origin are currently outside the scope of the regulation.
This does not mean that these sectors will never be covered – but for now, the focus is on categories such as textiles, batteries, electronics, furniture and construction products.
The Digital Product Passport is part of the European Union's vision for a circular economy and product traceability throughout their entire lifecycle.
Every product that falls under the new Ecodesign for Sustainable Products Regulation (ESPR) will need to have a unique digital identifier accessible via a QR code or NFC chip.
The passport will contain data about:
Food products are explicitly excluded from the scope of ESPR.
According to Article 2 of Regulation (EC) No 178/2002, "food" means any substance or product intended for human consumption.
Reason: food already has its own strictly regulated framework for safety and traceability (Regulation (EC) No 178/2002 and subsequent).
Animal feed also falls under separate legislation.
The food and feed regulation provides a specific traceability system, so ESPR does not duplicate it.
Human medicines, defined under Directive 2001/83/EC, are outside the scope.
This sector already has unique traceability systems (such as the EU Falsified Medicines Directive, eLeaflet, etc.), so DPP does not apply.
Regulation (EU) 2019/6 governs veterinary medicines.
They have their own rules for safety, labelling and traceability.
Living organisms are not "products" within the meaning of ESPR and cannot have a DPP.
Reason: lack of standardised sustainability and lifecycle data in this context.
Includes tissues, organs and biomaterials used for medical purposes.
Excluded on ethical and regulatory grounds.
These exclusions should not mislead manufacturers and traders.
ESPR is a "framework" regulation – it will expand gradually through delegated acts that may cover new categories.
Even if your product is not yet covered:
All EU member states will apply ESPR in full through national legislation.
Manufacturers and importers will need to:
DPP will not apply to all products, at least not immediately.
But the trend is clear – towards full digitalisation and market transparency.
Even sectors that are excluded today (such as food or medicines) may be partially covered in the future when the regulator creates an appropriate framework.
Follow the news around ESPR and delegated acts to be prepared for the next steps of the European green transformation.
Where and How Will It Be Applied?
DPP Across Industries

DPP for Textiles
DPP provides traceability from fibre to recycling, proves brands' sustainability, and inspires consumer confidence.
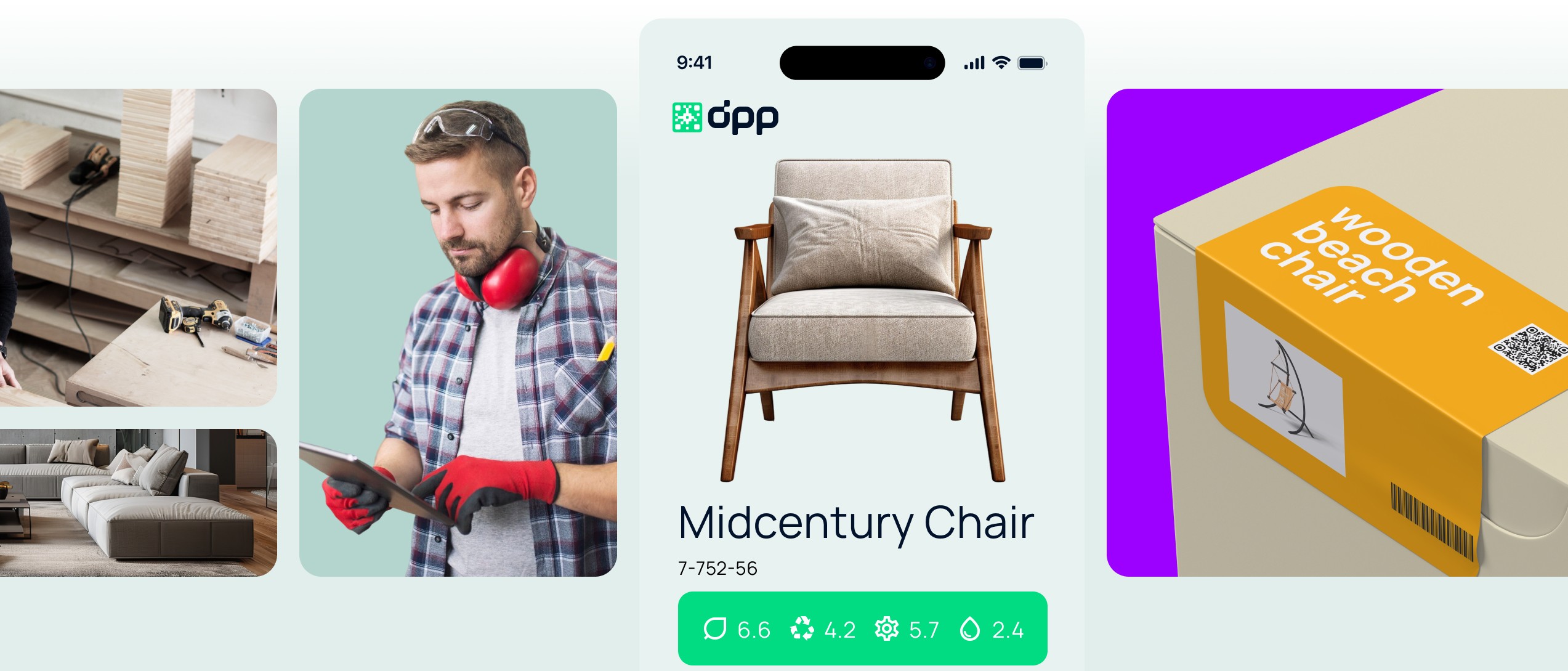
DPP for the Furniture Industry
DPP ensures transparency for the materials used, facilitates reuse and recycling. It proves the sustainability of production.
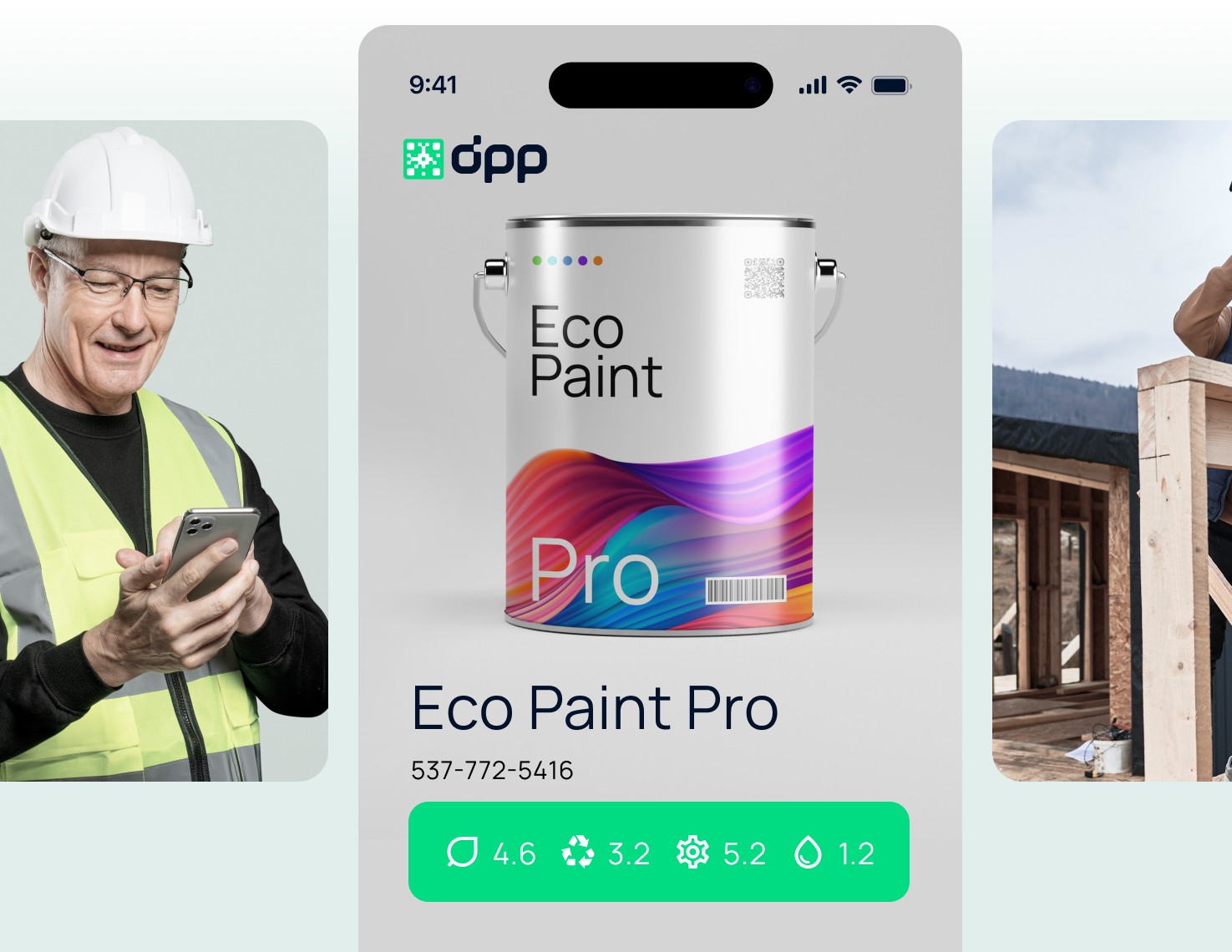
DPP for Construction Products
DPP ensures transparency for the composition and sustainability of construction materials, facilitating proof of origin and compliance with standards.
DPP in Business
Frequently Asked Questions
No, food is excluded from the scope of ESPR. It already falls under separate European regulations for food safety and labelling (such as Regulation (EC) No 178/2002). In the future, related food sustainability initiatives may be considered, but this will be outside the framework of DPP.
The reason is that for a number of categories – such as medicines, animal feed and food – specialised control, safety and traceability systems already exist. DPP is aimed at industrial, durable and physical products where sustainability and recycling are key factors.
No. Living organisms are not considered "products" within the meaning of ESPR. The regulation focuses on material goods produced for the purpose of placing on the market, not on biological entities.
Not at this time. They fall under Regulation (EU) 2019/6 for veterinary medicinal products. This framework already requires traceability and quality control, so duplication through DPP is not necessary.
Yes, and this is actually a strategic advantage. Voluntary DPP adoption demonstrates transparency and commitment to sustainability – factors that are increasingly valued by partners and customers. In some sectors, this may accelerate future regulatory compliance.
The first categories required to introduce a Digital Product Passport are associated with a high environmental footprint and complex manufacturing chains. The European Commission puts textiles, batteries, electronics, furniture and construction products at the top of the list. These sectors generate the most waste and have significant potential for reuse and recycling. With the introduction of DPP for them, the EU aims to improve market traceability, transparency and sustainability, gradually adding other product groups in the coming years.
Latest DPP Articles

DPP in cosmetics: The end of greenwashing and the beginning of conscious beauty

The EU Battery Passport: How the DPP Is Transforming the Energy Transition From the Mines to Recycling

End of "Greenwashing": The Digital Passport Revolution

DPP in construction: The new rules

DPP in Education: The Path to Sustainable Development

Digital Product Passport Operator: The profession that is changing the industry

How does the DPP change the rules for non-EU providers?

Contact us and learn all the details about its implementation

Get Started
Schedule a meetingHome
About Us
ESPR - Core Framework
DPP - Digital Product Passport
Work Plan 2025-2030
DPP - First Affected Industries
© 2026 WIARA DAAS Ltd.
All rights reserved.